As of March 10, 2021, more than 95 million doses of the COVID-19 vaccine had been administered across the U.S. And, as jabs went into arms, misinformation regarding the immunization continued to spread across the internet.
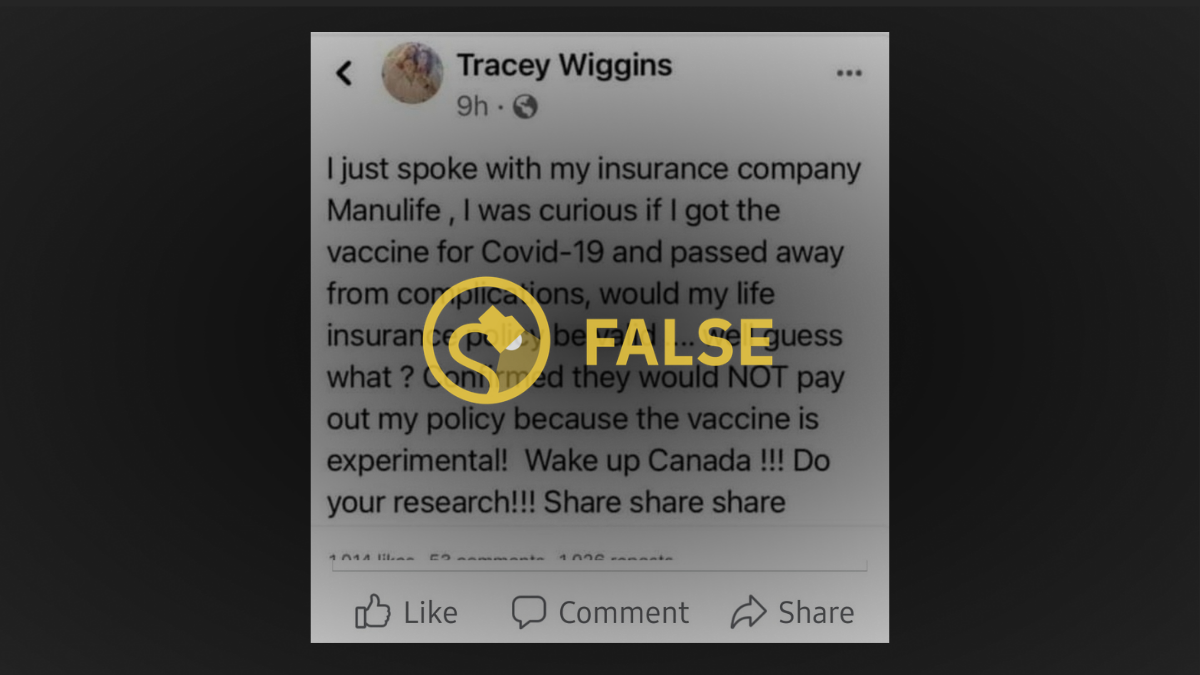
A number of social media users argued in March 2021 that life insurance providers could deny payout of a death benefit to beneficiaries if the policyholder died as a result of an “experimental” COVID-19 vaccination.
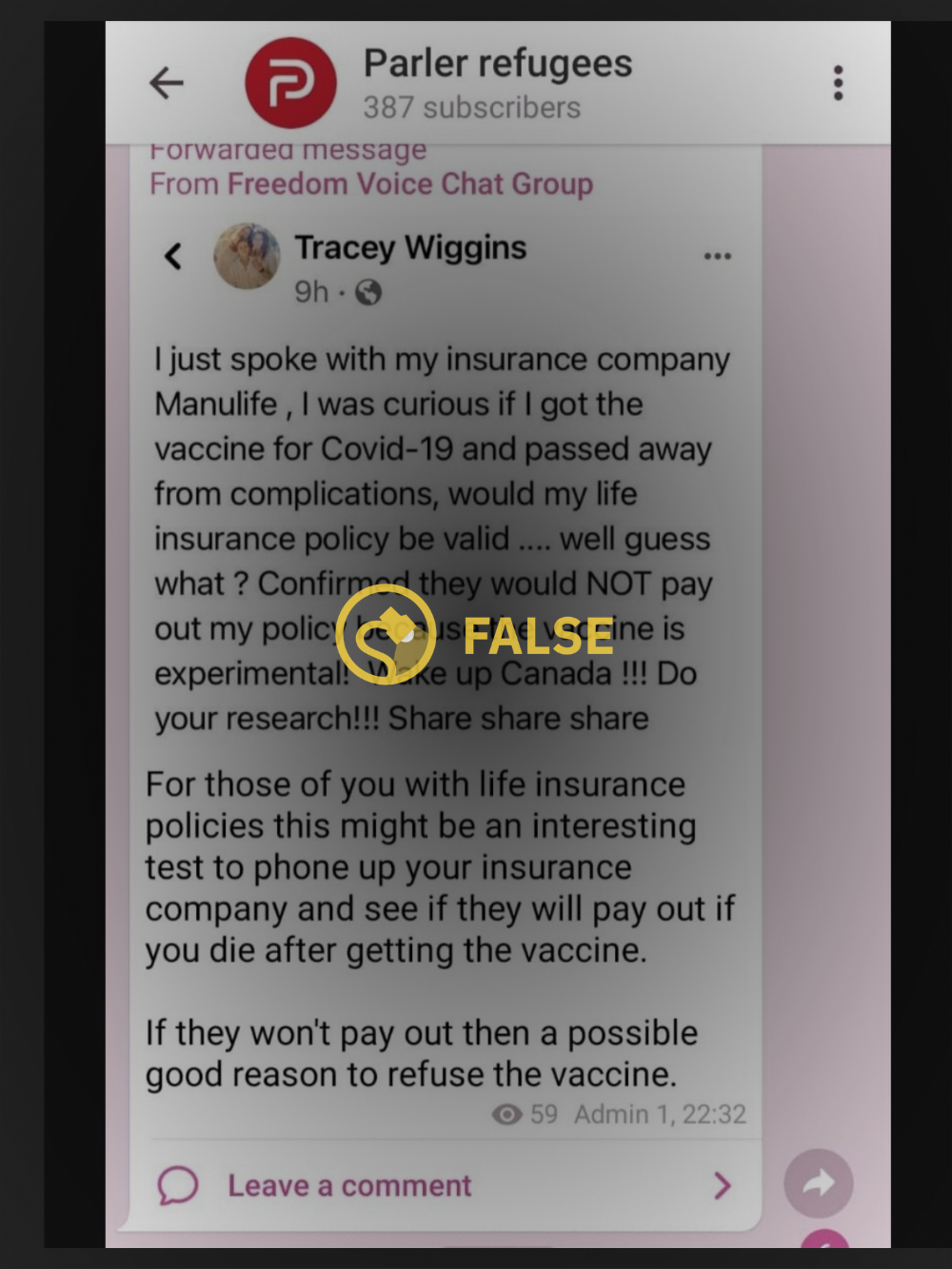
The claim spread widely on Twitter but appeared to have originated on an unidentified social media platform shared in a group called Parler Refugees, presumably a forum for those who previously used Parler, a social media platform popular among the alt-right and conspiracy theorists.
This claim is false.
The original post was shared by a user by the name of Tracey Wiggins, and was specific to Canada. In response to the misinformation being shared online, the Canadian Life and Health Insurance Association, Inc. (CHLIA) issued a statement on March 8 declaring that receiving the COVID-19 vaccine will have no effect on individual or workplace coverage or benefits from life insurance or supplementary health insurance, including disability, nor will it have any effect on the ability to apply for future coverage.
“No one should be afraid and choose to not protect themselves from COVID-19 because they are worried about it affecting their benefits,” said Stephen Frank, President and CEO of CLHIA. “All of Canada’s life and health insurers are supportive of Canadians receiving government-approved vaccinations to protect themselves from serious illness and death.”
The insurance lobbying group American Council of Life Insurers (ACLI) spokesperson Jack Dolan also confirmed to Snopes in an email that receiving the COVID-19 immunization will have no effect on life insurance in the U.S. — and even in instances of experimental drugs, life insurers have to pay death claims.
At the time of writing, three mRNA vaccines individually manufactured by Johnson & Johnson, Moderna and Pfizer-BioNTech were being distributed in the U.S. All three vaccinations were approved by the U.S. Food and Drug Administration under an Emergency Use Authorization, which allows for new therapeutics to be administered during public health emergencies like the coronavirus pandemic. An experimental or investigational drug, on the other hand, is one that has not received approval from government regulatory authorities but has shown promise in treating a disease or medical condition, according to the FDA. Under this definition, the COVID-19 vaccine would not be considered an "experimental" drug.
While COVID-19 vaccines are still being rigorously tested in broader-scale trials, health officials argue that the general safety and efficacy of the immunizations have warranted large-scale distribution. Snopes contacted the FDA and the National Institutes of Health for further clarification on the status of the three vaccines but did not receive a response in time for publication. We will update the article accordingly.
As of March 11, the Centers for Disease Control and Prevention Vaccine Adverse Event Reporting System (VAERS), has not detected patterns in the cause of death that would indicate a safety problem with COVID-19 vaccines.
"Over 92 million doses of COVID-19 vaccines were administered in the United States from December 14, 2020, through March 8, 2021. During this time, VAERS received 1,637 reports of death (0.0018%) among people who received a COVID-19 vaccine," wrote the agency.
"A review of available clinical information including death certificates, autopsy, and medical records revealed no evidence that vaccination contributed to patient deaths."
In some rare cases, health experts noted that some people experienced a severe allergic reaction known as anaphylaxis after getting their COVID-19 vaccine — approximately two to five people per 1 million in the U.S. However, reports indicate that anaphylaxis nearly always occurs within 30 minutes of receiving the vaccination and administrators will observe the vaccine recipient during this timeframe in the event that they need to respond.